 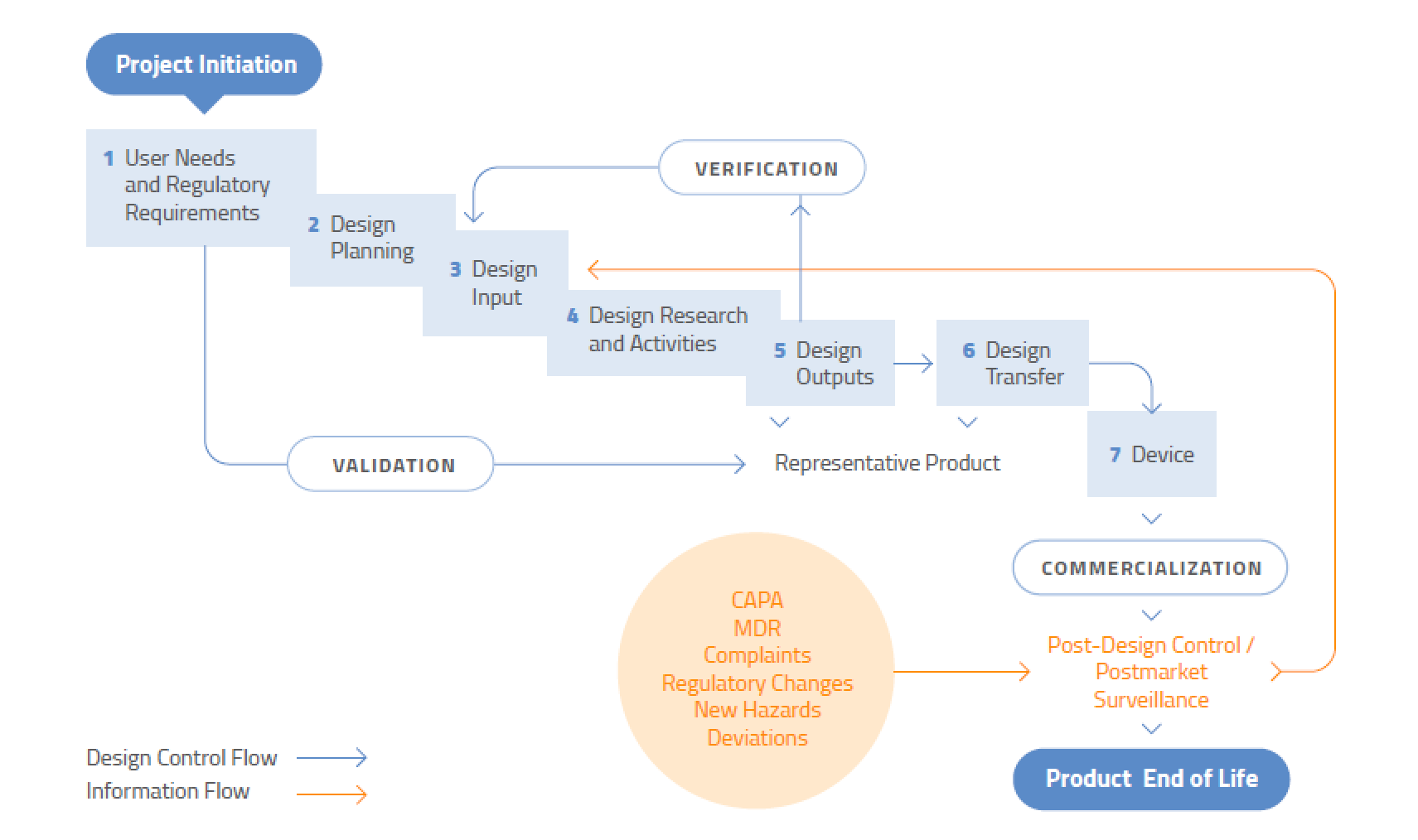
However, there are some requirements that might not be included explicitly in ISO 13485, for example Device History Record (FDA Part 820.184). Manufacturers can use ISO 13485:2016 for FDA 21 CFR Part 820 complianceīecause the FDA was instrumental in the revision of ISO 13485, most of the Part 820 regulation requirements are covered in ISO 13485. 
The FDA and other countries’ device regulatory agencies can more easily use and discuss inspection reports because the requirements are similar. The FDA played its part in the revision of ISO 13485:2016, because it is more convenient for industries to develop a Quality Management System if the needs of different countries are same. Numerous countries depend on ISO 13485:2016 in regulating medical devices. The FDA’s Quality System Regulation Part 820 is aligned with ISO 13485:2016 to a greater extent than ISO 9001:2015. The below comparison matrix will help you understand the working scopes, applications, and domains of both the standard and the regulation. 
ISO 13485 and FDA 21 CFR Part 820 are compared on the basis of their purposes, histories, scopes, and influences on each other. Other Parts include (for example) Part 810, which deals specifically with the procedure of medical device recall, and Part 830, dealing with unique device identification of medical devices. It is more similar to ISO 13485 as far as requirements are concerned. Part 820 defines requirements for the quality system to meet FDA regulations, termed current good manufacturing practices. Based on their compliance with this regulation, organizations can market medical devices commercially in the U.S. Organizations based on this standard can move towards compliance with FDA 21 Code of Federal Regulation (CFR) Part 820 (Quality System Regulation). The latest ISO 13485 standard also encompasses common regulatory concepts within its requirements. Relationship between ISO 13485:2016 and FDA 21 CFR Part 820įigure 1 depicts how ISO 13485:2016 helps organizations to maintain an effective Quality Management System that addresses the applicable regulatory requirements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
